 So it's going to bounce up and we call this the excited state, we have excited now the atom. Okay so this electron is going to bounce up to higher energy levels even though those energy levels are not necessarily occupied they are present in every single atom. so we're going to put energy into it whether it be like through electricity or through fire or through whatever type of energy that you want to put into it, that electron is going to bounce up. One red, one teal color and a few purple and blue.Īlright so what exactly is happening and why is this happening? Okay so we go over here ground state element in a ground state picture of hydrogen the electron is in it's lowest orbital in this case n equals 1, and if you pump energy into it, this electron is going to get what we call "excited" and it's going to go up to higher energy levels as you can see over here. And if we put a prism or a diffraction grading up to this light we're going to get a unique spectrum with these 5 bands of light. So the electrons are going to be excited and we'll talk about what excited means in just a second. This hydrogen and lamp we have a gas of hydrogen and obviously electricity is being put into it. We take hydrogen, which is as simple as atom and we're going to take, this is the hydrogen and the lamp it is inside of. So this actually are called finger prints of each element, and we can actually determine taking a light and putting a prism up to it or what we also call diffraction grading up to the light we can actually break it up and to tell what element is actually being excited or put energy in. So if you took a lithium light and broke it up you would see this just these bands within an electromagnetic spectrum and notice each element has its unique spectrum that it's going to emit. Instead of seeing the whole visible region like you do up here you would see something broken up. And finally potassium with the purple, or lilac shade.Īlright so if you took a prism to those actual the frame tests from those elements being burned or those compounds being burned. Then we have copper with its green color. Next, we have sodium giving us a characteristic yellow flame. Not showing up very well here in the next one, but the red tints inside of that orange, there's some impurities there that is lithium. 
Then we have boron being green, strontium giving us the crimson red color. Then we have some barium chloride, with barium ions burning a dirty yellow color. So let's actually look at the video that shows this kind of information.Ī very dramatic version of the flame test demonstration here. So what happens when you have light emitted from different elements than when you burn them or when you actually light something that's actually not white light when you break kit up with a prism what that actually looks like. Alright so we know that when you take white light put it to a prism it breaks up into the rainbow the full visible spectrum. 
In the middle we have visible light and we're going to focus on visible light just for next couple of seconds. 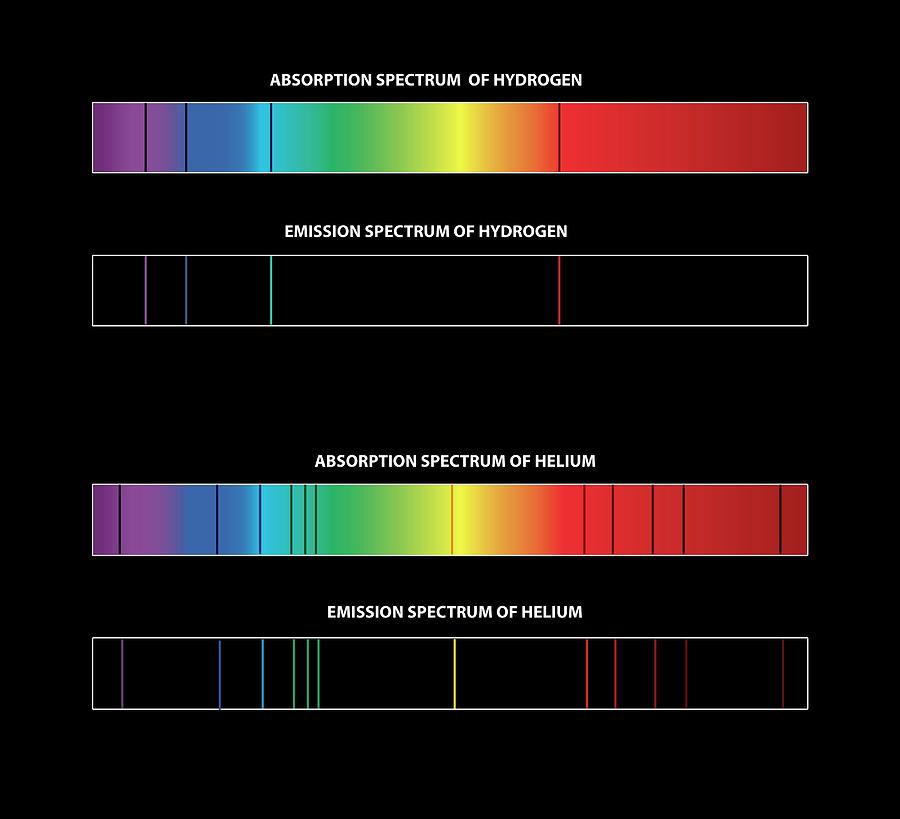 
Alright so here is the overall general view of the electromagnetic spectrum going from high energy wave the gamma rays to very large energy waves the radio wave. Okay so first question is electromagnetic spectrum let's take a look at that. It's a set of frequencies of the electromagnetic spectrum emitted by excited elements of an atom. Alright so we're going to talk about atomic emission spectra. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
